Dalton’s theory was presented by the John Dalton who was an English Chemist and had Quaker background. He is well-known due to his atomic theory. His fascination with atmosphere and weather led his research interest into nature of gasses which became foundation of atomic theory.
This theory has proposed that all of the matter was composed of the atoms, which are indestructible, and indivisible building blocks. Quickly, Dalton’s theory became a theoretical foundation in chemistry.
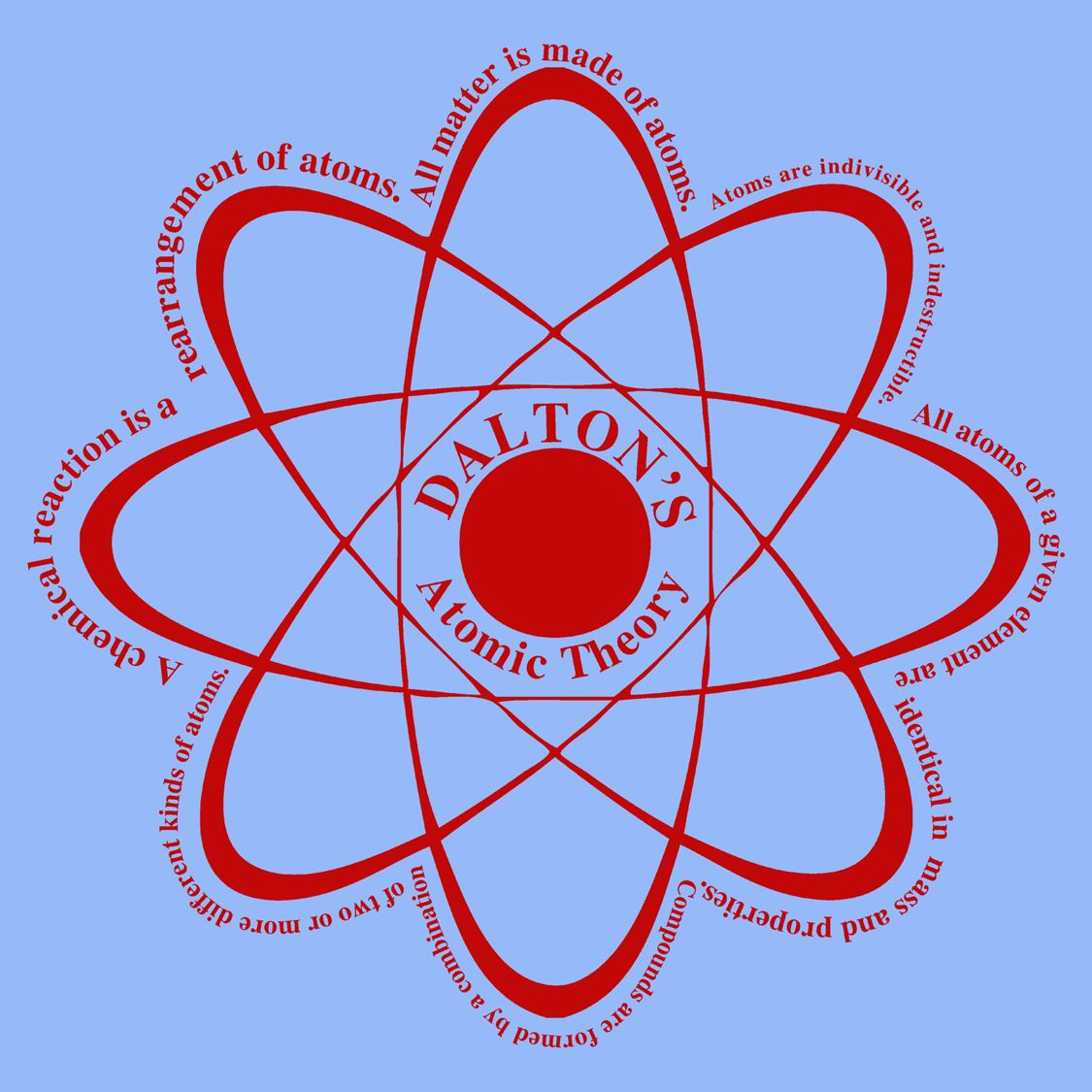
Mainly the points of Dalton’s atomic theory are as follows.
- All things are made up of atoms, which cannot be destroyed and is indivisible and significant building block of matter.
- Atoms are identical for each element.
- There is variation in mass and size of atoms of the different elements.
- Different whole number combination of the elements makes compounds.
- Atoms are rearranged as a result of chemical reaction and this rearrangement occurs in the compounds of reactants and products.
Few of the terms are also associated with Dalton’s atomic theory. According to that a standard unit that is atomic mass unit is used for indicating the mass of an atom. Atom is the smallest possible form of the matter whose identity is retained as chemical element. It was not known at that time that atom has nucleus that is surrounded by the electrons.
Merits of Dalton’s Atomic Theory
This theory explains laws of chemical combination such as law of multiple proportions and law of constant composition. He was the first person who recognized workable distinction between the fundamental particles of elements and compounds.
Drawbacks of Dalton’s Atomic Theory
- He stated that atom is indivisible but later it was proved wrong. Atom can be divided into neutrons, electrons, and protons. However, atom is smallest particle taking part in the chemical reactions.
- According to his theory for same element, atoms are similar in all the respects. But practically, atoms of several elements differ in their masses and densities and these atoms having different masses are called isotopes. For example, there are two isotopes of chlorine and their mass numbers are 35 and 37.
- This theory says that the atoms of two different elements differ in size, mass, and many other physical and chemical properties. However, it is not true for all the situations. For example, Calcium and Argon have atomic mass of 40 amu and are known as isobars.
- This theory could not explain the existence of allotropes.
However, some postulates of Dalton’s atomic theory are still valid and is a significant stepping stone in chemistry and forms base for quantum mechanics and modern atomic theories.
Of course, the modern atomic theory is more involved and modernized than the Dalton’s atomic theory but the essence of Dalton’s atomic theory is still valid. Dalton has also explained many unexplained chemical phenomena with his theory. This theory has been revised over many years, for the interconversion of mass and energy to incorporate existence of the atomic isotopes. Now new discoveries have been made and atom can be divided to the smaller parts. However, Dalton’s atomic theory has own significance and it has been recognized as the atomic mass unit as Dalton is designated to acknowledge its worth.